What is Disinfectant Efficacy Testing?
Your company applies disinfectants, sporicides, and sanitizers to product contact and facility surfaces, hoping to control microbial bioburden and to prevent product contamination. But is hope really a strategy? Is it working? Are the materials and processes validated?
We aren’t talking about cleaning validations. Cleaning validations confirm that the process doesn’t leave product or cleaning residue behind, and that any microbes which might have been residing on your surfaces are knocked down.
We aren’t talking about Environmental Monitoring (EM) either. This is an important tool that confirms random spots in your facility are microbially acceptable on an ongoing basis.
But will your antimicrobial program actually kill elevated quantities of the microbes living in your facility? Will it kill them if they are lurking on your product contact and facility surfaces? And what about your procedure? Are the steps outlined for antimicrobial preparation, application, and contact time adequate? Disinfectant efficacy testing is a stress test, marrying elements of your EM program, cleaning validation, and cleaning process into a single verification test to confirm that you will be protected from the inevitable microbial attack.
Antimicrobials manufactured for solid surfaces are regulated by the Environmental Protection Agency under the Federal Insecticide Fungicide and Rodenticide Act (EPA FIFRA). Products are registered with the EPA after significant GLP testing. Part of this testing of course confirms antimicrobial efficacy. While rigorous, this testing doesn’t necessarily reflect what you want the product to do in your facility. Disinfectants are registered for the EPA via the AOAC Use Dilution test. Briefly, P aeruginosa and S. aureus are deposited on stainless steel cylinders and allowed to dry. The cylinders are then soaked in the use dilution of the disinfectant for ten minutes, at which time they are retrieved and placed in a neutralizing growth medium. No growth should result from any of 180 replicates for it to be registered. While a rigorous test, it is not reflective of what’s happening in your facility. Are all of your surfaces highly polished stainless steel? Is that ATCC strain of S. aureus as resistant as the Micrococcus luteus your compounding technician is shedding into the space? Is your cleaning crew properly applying this product? And for the correct contact time? And how long is the use dilution stable for? Can you leave it for a week in that bottle and still expect it to be effective?
And if because it’s the right thing to do isn’t enough reason to perform this testing, this is a commonly found deficiency noted on US FDA 483 Audit Findings and Warning Letters. See the end of this blog for some links to enforcement actions.
How does Disinfectant Efficacy Testing Work?
FOCUS Laboratories has a library of coupon materials that reflect your manufacturing environment (stainless steel, tempered glass, latex gloves, epoxy floors). We utilize coupons that are about 2 x 2 inches square.
We typically enlist the usual suspects, the five USP test species, to cover the different groups of objectionable microbes:
- Staphylococcus aureus: Gram positive cocci, commonly found on humans, causes toxic shock, food poisoning, skin infections.
- Pseudomonas aeruginosa: Gram negative rod, skin and lung pathogen, commonly found in water and soil environments.
- Bacillus subtilis Gram positive spore forming rod. The spores are resistant to disinfection.
- Candida albicans: A fungal yeast, causes thrush.
- Aspergillus brasiliensis: A filamentous fungus.
But then importantly, we will use a few microbes that we have actually isolated from your facility during your environmental monitoring program. These are robust flora that haven’t spent generations on rich tryptic soy agar like the “usual suspects” listed above. These are wild strains.
We dilute the wild strains to reasonable levels, spread them on the coupons, dry them for a brief period of time, follow your procedure on how to clean and disinfect that surface, and then recover any survivors.
These studies can become quite large very quickly. Imagine:
- 5 surfaces
- 3 antimicrobials
- 8 test species
- 1 contact time
- 3 replicates
When totaled up the laboratory has 360 separate tests to perform. And that does not include demonstration of neutralization (so we are sure our contact time count is accurate) and initial counts. FOCUS has worked out a system that allows us to wrap up these projects quickly and accurately. You should be selective in how you pick a laboratory to perform this test. There are several considerations that a lab who doesn’t have the experience will not adequately address. These include:
- Drying time: How to dry the microbe on the surface without killing it. We have a unique method that varies for each test species.
- Antimicrobial application: It should mimic what is actually being performed in your facility. We will need to discuss use dilution, contact time, application mechanism, rinse, etc.
- Organism recovery: What’s the best way to get the microbes back off the coupon? Swab? Rinse? Contact plate? We have found our unique rinsing method to offer the best recovery rate. We will share the results of that study in a future blog.
- Disinfectant neutralization: Will the lab show complete kill because they haven’t neutralized the disinfectant and its still killing the microbes in their petri plates? Do they have data demonstrating antimicrobial neutralization? Was it performed properly?
FOCUS is a cGMP facility, so naturally we have validated our own disinfectant products, surfaces, and processes here are some of the test data for sterile 70% IPA:
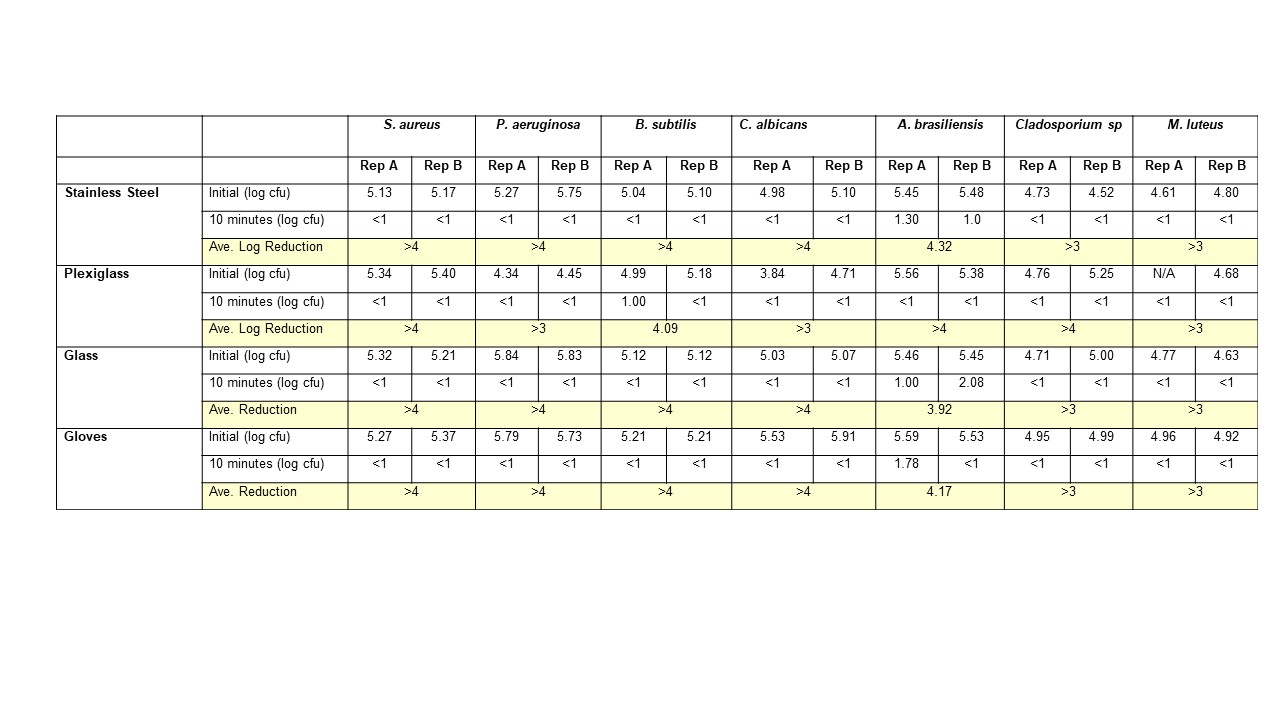
Note the lack of incomplete kill of the fungus from these results. This is why we do a daily wipe down with an EPA disinfectant product, and a weekly clean with an EPA Sterilant. And why we have data to prove efficacy.
USP <1092> Disinfectants and Antiseptics offers more detail on why this testing is necessary and additional factors to consider. And the internet gives plenty of examples of regulatory action taken against firms who haven’t demonstrated disinfectant efficacy:
Send us an email at info@focus-lab.com or give us a call at (610) 866-7272. We want to discuss how we can get this done for you — quickly and affordably. We would be happy to provide you with a quote and a free protocol.

